- Review
- Open access
- Published:
PARP inhibitors in gastric cancer: beacon of hope
Journal of Experimental & Clinical Cancer Research volume 40, Article number: 211 (2021)
Abstract
Defects in the DNA damage response (DDR) can lead to genome instability, producing mutations or aberrations that promote the development and progression of cancer. But it also confers such cells vulnerable to cell death when they inhibit DNA damage repair. Poly (ADP-ribose) polymerase (PARP) plays a central role in many cellular processes, including DNA repair, replication, and transcription. PARP induces the occurrence of poly (ADP-ribosylation) (PARylation) when DNA single strand breaks (SSB) occur. PARP and various proteins can interact directly or indirectly through PARylation to regulate DNA repair. Inhibitors that directly target PARP have been found to block the SSB repair pathway, triggering homologous recombination deficiency (HRD) cancers to form synthetic lethal concepts that represent an anticancer strategy. It has therefore been investigated in many cancer types for more effective anti-cancer strategies, including gastric cancer (GC). This review describes the antitumor mechanisms of PARP inhibitors (PARPis), and the preclinical and clinical progress of PARPis as monotherapy and combination therapy in GC.
Background
Gastric cancer (GC) is one of the most common cancers worldwide and the second leading cause of cancer-related deaths [1]. Due to its high incidence, high mortality rate, and extremely poor prognosis, it poses a serious threat to human health and life. Currently, surgical resection is the best treatment for patients with early GC [2], and chemotherapy is the most important treatment for patients who cannot undergo surgical resection or have advanced metastases [3]. However, GC is biologically and genetically highly heterogeneous [4], resulting in less than optimal results from surgical resection and chemotherapy. It is therefore urgent to explore more effective therapeutic strategies. The idea that DNA damage-deficient mechanisms promote tumorigenesis was recognised 20 years ago [5], and direct or indirect targeting of DNA damage repair pathways had emerged in cancer treatment approaches, and the clinical success of PARP inhibitors (PARPis) had made “synthetic lethal” anti-cancer therapies a new hope.
The replication fork encounters a lot of DNA damage during each cell cycle, and cells have developed several specific pathways to prevent specific DNA damage from causing cell “survival” problems [5, 6]. Among them, poly (ADP-ribose) polymerase (PARP) plays an irreplaceable role in the repair of DNA single-strand breaks (SSB) [7]. One of the most dramatic post-translational modifications caused by DNA damage is protein poly (ADP-ribosylation) (PARylation), catalyzed by members of the PARP nicotinamide adenine dinucleotide (NAD+) dependent ADP-ribosyltransferase superfamily [8], which is used to accelerate damage repair and plays a key role in cell fate determination. When SSB are not repaired after PARPis, it is transformed into DNA double Strand Breaks (DSB) in the S phase. DSB is considered one of the most lethal forms of DNA damage [9]. In normal cells, DSB can be repaired by homologous recombination repair (HRR). However, in cancers with homologous recombination deficiency (HRD), DSB can only be repaired with low-fidelity forms of DNA repair (e.g. Non-homologous end joining, NHEJ) after the use of PARPis, which will lead to a significant increase in genomic instability and cell death due to apoptosis or mitotic catastrophe [10]. This “synthetic lethality” occurs when there is a strong lethal synergy between two non-lethal events [11].
HRD related genotypes have been found in GC for many years [12]. To date, PARPis have been used as “gold finger” drugs in various cancer therapies, which has made them a focus of GC research. Nowadays, the efficacy of PARPis has been validated in various clinical trials in GC. Therefore, this review will first briefly describe the antitumor mechanisms of PARPis and then outline the preclinical and clinical progress of PARPis as monotherapy and combination therapy in GC.
The mechanism of PARPis
ADP-ribosylation
One of the most common tools to induce a rapid change in the cellular environment is the post-translational modification (PTM) of proteins by addition of chemical moieties, such as phosphate, acyl (most commonly methyl and acetate), small proteins or sugars [13]. Of these, the least studied of the various histone modifications is likely to be ADP-ribosylation. ADP-ribosylation also is a PTM, in which ADP-ribosyltransferases use NAD+ to modify target proteins with ADP-ribose. This modification, occurring as mono- or poly-ADP-ribosylation [14], can alter the physical and chemical properties of target proteins [15]. ADP-ribosylation is associated with many cellular processes, including different forms of stress and metabolism, such as DNA repair, transcription, regulation of meristem function, telomere length and senescence, protein degradation, apoptosis and necrosis [16,17,18]. This may be due to the fact that poly (ADP-ribosylation) is usually associated with the relaxation of chromatin structure in target proteins.
The enzymes involved in ADP-ribosylation are mainly those of the ART (ADP-ribosyltransferase) superfamily, characterized by structural homology of the ART domain to either diphtheria toxin (ART diphtheria toxin like, ARTD) or cholera toxin (ART cholera toxin like, ARTC). In mammals, the only known proteins that can be poly (ADP-ribosyl) ed. are members of the PARP family [19]. Thus, PARPs can be considered as ADP-ribose “writers” (writers being the enzymes that catalyse the formation of ADP-ribose modifications), which attach (“write”) ADP-ribose units to substrate proteins in a covalent manner. Removal of the ADP-ribose chain is catalyzed by “erasers” including PAR glycolytic enzymes (PARG), ADP-ribosyl hydrolase 3 (ARH3), TARG and MacroD1/D2 [20, 21] (Fig. 1).
Regulation of reversible ADP-ribosylation. ADP-ribosylation is a post-translational modification of the protein, the use of NAD+ as a donor, PARPs act as synthases to ligate ADP-ribose to target proteins, ADP-ribose is also a reversible process, ADP-ribose can be removed by hydrolytic enzymes, such as PARG, ARH3 and so on
The family of PARP
The PARP protein family consists of 17 members that have distinct structural domains, activities, subcellular localizations, and functions [22, 23]. However, they share a conserved catalytic PARP domain fold that is homologous to the diphtheria toxin ART fold and is therefore also known as diphtheria toxin-like ADP-ribosyltransferase (ARTDs) [8, 24]. Because poly (ADP-ribosyl) isomerization is often associated with the relaxation of chromatin structure, PARP may play a central role in many cellular processes, including DNA repair, replication, and transcription. Based on their structural domains and functions, the different PARPs can be broadly classified as DNA-dependent PARPs (PARP1, PARP2, and PARP3), Tankyrases (PARP5a and PARP5b), Cys-Cys-Cys-His zinc finger (CCCH)-containing and WWE PAR-binding domain-containing PARPs (PARP7, PARP12, PARP13.1, and PARP13.2), and PAR-binding macrodomain-containing “macro” PARPs (PARP9, PARP14, and PARP15). The remaining PARPs are referred to as unclassified PARPs [20, 23] (Table 1).
PARP involvement in the cellular response to DNA damage has long been appreciated and continues to actively develop [25, 26]. PARP, a sensor of DNA damage, coordinates DNA repair processes by providing poly-ADP-ribose chains that function as interaction platforms for repair and signaling proteins [14]. The extent of the PARylation response to DNA damage largely depends on the nature and amount of DNA breaks produced. In response to low levels of DNA lesions, PARP activity favors repair and survival. In the presence of extensive DNA injury as observed during ischemia/reperfusion and inflammatory conditions, the massive production of PAR ultimately causes cell-death via at least two distinct mechanisms: energy-failure induced necrosis or apoptosis-inducing factor (AIF) dependent apoptosis [27]. A tight regulation of PARylation homeostasis is therefore of critical importance for efficient repair when cells are exposed to sub-lethal doses of DNA damage [28]. PARP activation consumes NAD+ and alters AMP:ATP ratios through the actions of PARG, which hydrolyses pADPr into ADP-ribose, and of the ADP-ribose pyrophosphohydrolase NUDIX enzymes, which cleave ADP-ribose into AMP and phospho-ribose [29].
PARP in cancer
Defective DNA repair is a common hallmark of cancer [30]. Compared with normal cells, cancer DDR is characterized by losing one or more DDR pathway or capability during their generation, leading to a greater dependency on the remaining pathways [31, 32]. The DDR is a cellular response to exogenous and endogenous genotoxic damage that may result in SSB and DSB. Of these, DSB are considered one of the most lethal forms of DNA damage [33]. In normal cells, the accumulations of DSB are repaired preferentially by HR rather than NHEJ [34, 35]. HR is a high-fidelity repair pathway that utilizes the sister copy of the damaged DNA as a template, leading to the reconstitution of the original sequence [36, 37]. In contrast, NHEJ is intrinsically error-prone, modifies the broken DNA ends, and ligates them together with little or no homology, generating deletions or insertions [35]. Tumor cells, lacking a functional HR pathway will be more dependent on SSB repair mechanisms. PARP itself constitutes a component of the base excision repair (BER) pathway [38]. It is well established that PARP proteins bind to SSB and that poly-ADP-ribose polymerase, as a cofactor, is required for the effective repair of SSB [39]. It accomplishes SSB repair by recruiting XRCC1 and ligase III to the site of injury [40,41,42]. Meanwhile, PARP contributes to DSB repair as a component of an alternative pathway of NHEJ [43]. PARP1 also contributes to HR system functioning by recruiting critical DNA repair factors such as NBS1 and MRE11 to sites of DSB and by preventing Ku70/80 proteins from binding to areas of DNA damage. Ku proteins are essential components of the error-prone NHEJ pathway, and PARP1 exerts an active role in its inhibition [44, 45]. PARP3 has newly been acknowledged facilitating the process of NHEJ [46]. Thus, PARP can inhibit classical non-homologous end joining (cNHEJ) to some extent, allowing PARP to be highly activated in HRD tumor cells [47].
“ Synthetic lethality “ of PARPis
Because different DNA-repair pathways can overlap in function, and as one pathway can sometimes ‘back-up’ for defects in another, inhibition of such pathways present in cancer cells could have a greater impact on the tumor tissues than normal tissues. Given the role of PARP in BER, inhibitors targeting PARP were developed in cancers with HRD to cause the accumulation of persistent SSB.
The main mechanism of PARPis involves the inhibition of PARP enzymatic activity (formation of poly ADP-ribose chains from NAD+), which is required for both relaxing chromatin and PARP dissociation from the DNA that occurs following auto-modification. Both of these events are required to facilitate SSB repair, and the structures of the PARPis are built around an NAD+ mimetic core. Consequently, competitive inhibition could substitute inactivated enzyme for NAD+ thus preventing NAD+ utilization on PARP protein, finally generating a potential block for cellular DNA replication [48]. The aforementioned process could trap PARP on the DNA, preventing auto-PARylation and PARP release from the site of the DNA lesion [49, 50]. Trapped PARP–DNA complexes were more cytotoxic than unrepaired SSB caused by PARP inactivation, arguing that PARPis act in part as poisons that trap PARP enzyme on DNA [50, 51]. At the same time, trapped PARP-DNA complexes can lead to the stalling and/or collapsing of replication forks, resulting in the generation of more deleterious DSB [52]. A second mechanism of PARP inhibitors demonstrates that PARP can inhibit cNHEJ [46]. As a result, PARPis will enhance NHEJ. NHEJ will result in a significant increase in genomic instability that over multiple rounds of replication will become unsustainable and result in tumor cell death [47]. Recently, a third mechanism for the sensitivity of PARPis has been discovered that PARPis decreases efficiency of mutagenic microhomology-mediated end joining (MMEJ)-based repair [53]. HR factors suppress MMEJ following DSB resection. Due to the defect in HR, tumor cells can rely on MMEJ (or Alt-EJ pathway) for the DSB repair pathway. This pathway is dependent on PARP and the trans-disease polymerase (POLQ). In fact, PARP is required for efficient recruitment of POLQ to DSB. Thus, inhibitors of PARP or POLQ will block the Alt-EJ pathway and kill HR-deficient tumor cells [53,54,55].
Although the precise mechanism by which PARPis kill tumor cells remains to be fully clarified, the anticancer effect is attributed to catalytic inhibition of PARP that block repair of SSB. While PARPis is well-tolerated by normal cells, this effect of PARPis is more likely observed in tumor cells with a HR- deficient background [48]. As a result of defective enzymatic function induced by PARPis, the accumulation of SSB is subsequently encountered by replication forks and generates potentially lethal DSB that need to be fixed [56]. In the absence of a functional HR pathway, highly specific PARPis induce DNA damage and tumor-restricted gene repair functions, resulting in the loss of required DNA repair functions and the death of tumor cells, leading to the concept of “synthetic lethality” [57, 58] (Fig. 2).
The principle of synthetic lethality. PARP is involved in SSB repair, and when PARP inhibitors can bind competitively with PARP, it leads to a large accumulation of SSB in the cell that cannot be repaired in time. Unrepaired SSB will lead to the collapse of replication fork, resulting in DSB. In normal cells, DSB can be repaired. In HRD tumor cells, DSB can not be repaired, resulting in a significant increase in the probability of tumor cell death. According to the synthetic lethal principle, PARP inhibitors can selectively kill tumor cells without affecting normal somatic cells
PARPis have demonstrated their clinical efficacy in HR-deficient cancer entities, for example, some of PARPis had been used clinically to treat breast cancers with BRCA 1 or 2 pure deletions [59, 60]. BRCA1 and BRCA2 are key components of the HR pathway. Initial observations were quickly expanded and it was demonstrated that defects in other HR components (e.g., RAD51, RPA1, ATM, ATR, CHK2, TOPBP1, ARID1A, etc.) were also associated with PARP inhibitor sensitivity [61,62,63]. Effect of PARPis may depend on the specific genetic environment of the specific tumor. For example, the survivals of BRCA1/2-deficient human breast tumors are dependent on the PARP/POLQ pathway, whereas the mechanisms corresponding to other mutations may be different [51].
There are some studies showing a survival benefit of PARPis in patients with tumors regardless of BRCA1/2 mutation status or HR repair status [64]. The findings indicate that PARPis treatment induces IFN-mediated antitumor immune responses. PARPis generate cytosolic dsDNA, which activate stimulator of IFN genes (STING) signaling and its associated transcription programs. These critical changes amplify STING signaling, and promote tumor-infiltrating lymphocytes (TIL) and antitumor immunity, which can be further enhanced through immune checkpoint blockade [65]. This makes us more aware of the deeper possibilities of the PARPis mechanism for better use of PARPis.
Drugs targeting PARP
Since 2014, three PARPis — olaparib, rucaparib and niraparib—have been approved by the Food and Drug Administration (FDA) and European Medicine Agency (EMA) for application in ovarian cancer [66]. For instance, olaparib and rucaparib have been approved to treat BRCA-defective prostate cancer patients [67]. The FDA announced the approval of talazoparib for the treatment of harmful or suspected harmful germline BRCA-mutant, HER2-negative locally advanced or metastatic breast cancer. Studies involving other PARPis have also shown encouraging clinical results and are likely to receive additional approvals in the near future, such as veliparib, pamiparib (BGB-290), fluzoparib, E7449 and IMP4297. We have classified them into Pan-PARPs as well as Specific PARPs depending on the target (Table 2). The majority of the PARPis identified to date are pan-PARPis that target multiple PARPs, and the variety of PARP inhibitors is remarkable. We searched the term “PARPi” on Clinicaltrial.gov and found 298 relevant studies, of which 75 clinical trials have been completed as a single drug or in combination with other therapies (https:// clinicaltrials. Gov //). There are up to a dozen types of PARPi drugs in clinical trials; there are also up to a dozen drugs in preclinical studies (Table 2).
PARPis in gastric Cancer
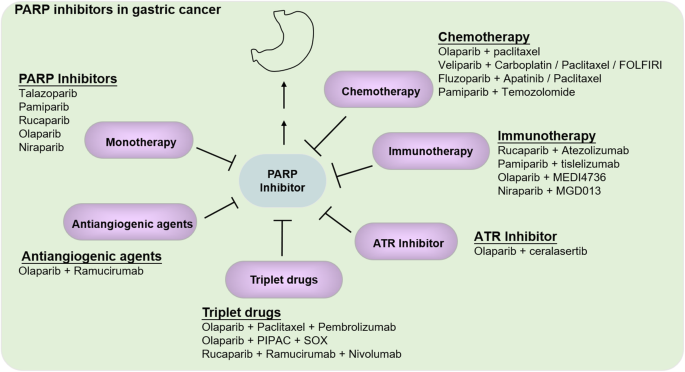
GC is one of the most common malignant tumors of the digestive system [1], which is a highly heterogeneous disease. Although emerging targeted strategies and immunotherapies have brought new hope to antitumor therapy, options for advanced GC with high heterogeneity are still few. Only three drugs have been currently approved (trastuzumab, lamocirumab, and apatinib), but the prognosis for advanced GC remains poor. Hence, the novel strategies are urgently needed for advanced GC [81]. The most recurrently mutated genes of GC, which were validated in a separate cohort of 216 cases by targeted sequencing, were members of the homologous recombination DNA repair, WNT and PI3K-ERBB pathways [82]. Deficiencies in HR are extensively seen in gastric cancer. Mutations in PALB2, BRCA1, BRCA2, ARID1A, ATM or RAD51C genes, which regulate homologous DNA recombination, were widely identified in GC [49, 82, 83]. Tumors that developed in patients with these mutations had a mutation signature associated with somatic HRD. Hence, based on the above description of PARPis function, it is enough to see that the application of PARPis in GC is promising. Here we will list some studies about single PARPis drugs investigated in GC either in the preclinical or clinical phase as follows (Fig. 3).
Monotherapy of PARPis
In the treatment of GC, PARPis has some non-DNA repair functions in addition to the above well-known functions of blocking DNA repair. Vascular endothelial growth factor (VEGF) and vascular endothelial growth factor receptor 2 (VEGFR2)-mediated signaling and angiogenesis can contribute to the pathogenesis and progression of GC [84]. PARPis cause loss of ERK2 stimulation by decreasing the activity of critical pro-angiogenic factors including VEGF, transmembrane signaling protein syndecan-4 (SDC-4) and hypoxia inducible factor (HIF). This ultimately results in reduced angiogenesis and inflammation [84,85,86]. Furthermore, inhibition of PARP1 is associated with transcriptional silencing through accumulation of DNA methylation and CpG island hypermethylation throughout the genome [87]. PARP1 can also hinder DNA methylation by dimerization with DNA (cytosine-5-)-methyltransferase 1 (DNMT1), a methyltransferase found overexpressed in gastrointestinal tract carcinomas, resulting in inhibition of its methyltransferase activity [87]. Recently, PARP1 inhibition has been addressed to attenuate the AKT-associated phosphorylation of forkhead box O (FOXO) transcription factors [88]. PARP1 is known to indirectly regulate FOXO3A via the NF-κB or AKT pathway and plays an important role in the progression of GC [89, 90].
Preclinical
In preclinical studies, GC cell lines showed sensitivity to olaparib, especially those with low ataxia telangiectasia mutant (ATM) protein expression or RAD51C-Deficient [91, 92]. The possible cause is the deficiency of HR pathway due to the loss of these proteins.
Clinical
Clinical trials about PARPis are ongoing. Olaparib was the first PARP inhibitor introduced in clinical practice [93]. A multicenter, non-randomized, phase II trial (NCT03829345) involving GC was recruiting, patients will be administered olaparib orally twice daily (BD) at 300 mg continuously for each 28 days cycle. Other single-drug phase II trials, including Talazoparib (NCT04550494), pamiparib (NCT03427814), Rucaparib (NCT04171700), Niraparib (NCT03840967) (Table 3), are also underway and the results are promising.
Combination therapies of PARPis
Targeting PARP has recently been identified as a highly promising option for targeting GC. However, individual PARPis have limited efficacy. Combination strategies are desirable both to optimize the efficacy of PARPis and to expand the population of benefit (Fig. 3).
Preclinical
Currently, PARPis are being studied in preclinical trials in synergy with a number of drugs in GC, several of which have been administered in combination therapy with encouraging results. Both as chemosensitizers or radiosensitizers, as well as in combination with other antineoplastic therapies. These preclinical trials provide a theoretical basis for the clinical application of synergistic regimens. In a preclinical study, the results demonstrated that combined treatment with PI3K and PARP inhibitors effectively inhibited the growth and migration of GC cells with ARID1A deficiency in vitro [94]. Moreover, because of the strong link between the efficacy of PARPis and impaired HR, a growing number of combination strategies with HR-deficient inducers have been developed for a variety of cancers [95,96,97]. In a recent study of GC, the study found that activation of c-MET increases tumor cell survival through the initiation of the DNA damage repair pathway. Herein, co-inhibition of c-MET and PARP enhances the levels of γ-H2AX and DNA damages in GC cells. The combinatorial treatment reduces the tumor and triggers apoptotic cell death growth in AGS xenograft models [98]. Furthermore, it is known from research that Checkpoint kinase 1 (Chk1) is a crucial regulator of cell cycle transition in DDR, it plays an important role in promoting the survival and growth of GC cells. Thus, combination with PARP1 inhibitor exhibited marked synergistic anticancer effect in both in vitro studies and in vivo experiments using a GC PDx model. Especially, Chk1 inhibitor combined with PARPis may be a more effective therapeutic strategy in GC. PARP inhibitor olaparib combined with WEE1/PLK1 dual inhibitor AZD1775 elicited potentiated anticancer activity through disrupting DDR signaling and the DNA damage checkpoint. AZD1775 abrogates olaparib-activated DNA damage checkpoint and causes mitotic DNA damage in GC cells. It sheds light on the combination strategy of WEE1/PLK1 dual inhibitors with PARPIs in the treatment of GC [81]. Based on these promising results, numerous clinical trials of combination strategies were conducted in GC patients. Table 3 summarizes these combinations of clinical trials that have been completed or are ongoing.
Clinical
PARPis and Chemoradiotherapy
Olaparib
PARPis were originally developed to enhance the antitumor activity of ionizing radiation and genotoxic agents [78, 99], they enhanced the efficacy of DNA alkylating agents, topoisomerase I poisons, and ionizing radiation [100]. Therefore, clinical trials (e.g. NCT01063517 and NCT01924533) have used this mechanism to show in more detail that this combination therapy shows a beneficial effect on patient survival. In a randomized, double-blind phase II clinical trial (NCT01063517), the combination of olaparib plus paclitaxel in patients with recurrent or metastatic GC with disease did not meet the primary endpoint of progression-free survival (overall population: hazard ratio [HR], 0.80; median PFS, 3.91 v 3.55 months, respectively; ATM low population: HR, 0.74; median PFS, 5.29 v 3.68 months, respectively). However, the combination of olaparib and paclitaxel was associated with a significant improvement in OAS versus paclitaxel plus placebo (13.1 vs. 8.3 months). This benefit was even more pronounced in patients with low ATM levels [101]. In the phase III GOLD trial (NCT01924533) clinical trial, olaparib/paclitaxel was generally well tolerated in both populations, however there was a trend in overall survival between the two populations but no statistical difference (median OS 8.8 months versus 6.9 months (HR = 0.79, p = .0262) [102]. Notably, ATM-negative patients treated with olaparib showed significantly improved remission rates compared to ATM-negative patients treated with paclitaxel alone (ORR 4.24, p = .0309), so PARPis may still be effective in selecting the correct biomarker for GC [103].
Veliparib
The results of A phase 1 dose-escalation study of veliparib with bimonthly FOLFIRI in patients with advanced solid tumors were also promising. Patients received veliparib (10–270 mg BID, days 1–5, 15–19) and FOLFIRI (days 1–3, 15–17) in three regimens containing 5-fluorouracil 2400 mg/m2: irinotecan 150 mg/m2 and folinic acid 400 mg/m2; irinotecan 180 mg/m2, folinic acid 400 mg/m2, and 5-fluorouracil 400 mg/m2 bolus, or irinotecan 180 mg/m2. Besides, ORR was 17.6% [104]. The acceptable safety profile and preliminary antitumor activity of veliparib plus FOLFIRI support further evaluation of this combination.
PARPis and targeted drugs
Anti-angiogenic therapies are known to induce a hypoxic cellular state that leads to downregulation of HR repair genes (BRCA1, BRCA2, and RAD51, among others), thereby enhancing PARPis sensitivity [105]. Furthermore, the studying of Olaparib and Ramucirumab in Treating Patients With Metastatic or Locally Recurrent Gastric or Gastroesophageal Junction Cancer That Cannot Be Removed by Surgery (NCT03008278) is ongoing. Of these, ramucirumab is a humanized monoclonal antibody that specifically blocks VEGFR2 and downstream angiogenesis-related pathways. And FDA has approved ramucirumab for the treatment of advanced gastric cancer or gastroesophageal union adenocarcinoma. AZD6738 is an orally active, selective ATR kinase inhibitor. The combination of AZD6738 and olaparib has received preclinical validation. A Phase I/II clinical study of ceralasertib in combination with olaparib (NCT02264678) is also currently underway and is highly anticipated.
PARPis and immune checkpoint inhibitors
In a Phase 1a / b clinical trial of pamiparib in combination with anti-PD-1tislelizumab in patients with advanced solid tumors (NCT02660034), 49 patients were enrolled (median age 63 years [IQR 55–67]), all of whom received at least one dose of pamiparib or tiselzumab. At a median follow-up of 8·3 months (IQR 4·8–12·8), ten (20%) of 49 patients achieved an objective response according to Response Evaluation Criteria in Solid Tumors (RECIST) version 1.1, including two complete responses and eight partial responses. And no grade 5 adverse events were reported. Pamiparib with tislelizumab was generally well tolerated and associated with antitumor responses and clinical benefit in patients with advanced solid tumors supporting further investigation of the combination of pamiparib with tislelizumab [106]. Equally, the phase 2 MEDIOLA basket trial assessed the efficacy and safety of chemo-free combination of olaparib and durvalumab in patients with solid tumors, including ovarian cancer, breast cancer and GC (NCT02734004). Combination of olaparib and durvalumab showed promising antitumor activity and safety similar to that previously observed in olaparib and durvalumab monotherapy studies. In GC, there are increasing clinical trials (NCT02264678, NCT04178460) underway that will help decipher the exact role of PARPis in combination with the anti-PD1 / PD-L1 strategy (Table 3).
Conclusions
Over the past few years, we have witnessed the use of PARPis in the treatment of GC and see a bright future in various preclinical and clinical data. However, because GC are highly heterogeneous tumors, PARPis may still be more effective in GC with specific biomarkers, such as the use of ATM loss as a predictive biomarker of GC response. FDA approval of Lynparza was accompanied by approval of a genetic test called BRACAnalysis CDx to detect the presence of BRCA gene mutations (gBRCAm) in blood samples from ovarian cancer patients [107]. We also expect that appropriate genetic assays will be available to monitor the reliability of PARPis for use in GC. A better understanding of PARP inhibitor resistance and the underlying mechanisms involved in its mechanism of action will also help in the development of new therapeutic strategies to address this issue [108]. Importantly, there is a need for novel adaptive trial designs to validate the various mechanistic hypotheses emerging in GC. By addressing these gaps in our knowledge, the use of PARPis in GC treatment will continue to expand as a “beacon of hope” in our anticancer armamentarium.
Availability of data and materials
Not applicable.
Abbreviations
- DDR:
-
DNA damage response
- PARP:
-
Poly (ADP-ribose) polymerase
- PARylation:
-
Poly (ADP-ribosylation)
- DSB:
-
DNA single strand breaks
- HR:
-
Homologous recombination
- GC:
-
Gastric cancer
- PARPis:
-
PARP inhibitors
- SSB:
-
Single-strand breaks
- HRR:
-
Homologous recombination repair
- HRD:
-
HRR deficiency
- PTM:
-
Post-translational modification
- NAD+:
-
Nicotinamide adenine dinucleotide
- PARG:
-
PAR glycolytic enzymes
- ARH3:
-
ADP-ribosyl hydrolase 3
- ARTDs:
-
Diphtheria toxin-like ADP-ribosyltransferase
- cNHEJ:
-
Classical non-homologous end joining
- CCCH:
-
Cys-Cys-Cys-His zinc finger
- AIF:
-
Apoptosis-inducing factor
- BER:
-
Base excision repair
- MMEJ:
-
Microhomology-mediated end joining
- POLQ:
-
Polymerase
- STING:
-
Stimulator of IFN genes
- TIL:
-
Tumor-infiltrating lymphocytes
- FDA:
-
The Food and Drug Administration
- EMA:
-
European Medicine Agency
- VEGF:
-
Vascular endothelial growth factor
- SDC-4:
-
Transmembrane signaling protein syndecan-4
- HIF:
-
Hypoxia inducible factor
- DNMT1:
-
DNA (cytosine-5-)-methyltransferase 1
- FOXO:
-
Forkhead box O
- ATM:
-
Ataxia telangiectasia mutant
- BD:
-
Orally twice daily
- RECIST:
-
Response Evaluation Criteria in Solid Tumors
- gBRCAm:
-
BRCA gene mutations
References
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68(6):394–424. https://doi.org/10.3322/caac.21492.
Katai H, Ishikawa T, Akazawa K, Isobe Y, Miyashiro I, Oda I, et al. Five-year survival analysis of surgically resected gastric cancer cases in Japan: a retrospective analysis of more than 100,000 patients from the nationwide registry of the Japanese gastric Cancer association (2001-2007). Gastric Cancer. 2018;21(1):144–54. https://doi.org/10.1007/s10120-017-0716-7.
Wagner AD, Syn NL, Moehler M, Grothe W, Yong WP, Tai BC, et al. Chemotherapy for advanced gastric cancer. Cochrane Database Syst Rev. 2017;8(8):Cd004064.
Padmanabhan N, Ushijima T, Tan P. How to stomach an epigenetic insult: the gastric cancer epigenome. Nat Rev Gastroenterol Hepatol. 2017;14(8):467–78. https://doi.org/10.1038/nrgastro.2017.53.
Hoeijmakers JH. Genome maintenance mechanisms for preventing cancer. Nature. 2001;411(6835):366–74. https://doi.org/10.1038/35077232.
Powell SN, Bindra RS. Targeting the DNA damage response for cancer therapy. DNA Repair (Amst). 2009;8(9):1153–65.
Pascal JM, Ellenberger T, et al. DNA Repair (Amst). 2015;32:10–6.
Amé J-C, Spenlehauer C, de Murcia G. The PARP superfamily. BioEssays. 2004;26(8):882–93.
Hoevenaar WHM, Janssen A, Quirindongo AI, Ma H, Klaasen SJ, Teixeira A, et al. Degree and site of chromosomal instability define its oncogenic potential. Nat Commun. 2020;11(1):1501. https://doi.org/10.1038/s41467-020-15279-9.
Ceccaldi R, Rondinelli B, D'Andrea AD. Repair pathway choices and consequences at the double-Strand break. Trends Cell Biol. 2016;26(1):52–64. https://doi.org/10.1016/j.tcb.2015.07.009.
Kaelin WG. The concept of synthetic lethality in the context of anticancer therapy. Nat Rev Cancer. 2005;5(9):689–98. https://doi.org/10.1038/nrc1691.
Ashworth A. A synthetic lethal therapeutic approach: poly (ADP) ribose polymerase inhibitors for the treatment of cancers deficient in DNA double-strand break repair. J Clin Oncol. 2008;26(22):3785–90. https://doi.org/10.1200/JCO.2008.16.0812.
Palazzo L, Mikoč A, Ahel I. ADP-ribosylation: new facets of an ancient modification. Febs J. 2017;284(18):2932–46.
Lüscher B, Bütepage M, Eckei L, Krieg S, Verheugd P, Shilton BH. ADP-Ribosylation, a multifaceted posttranslational modification involved in the control of cell physiology in health and disease. Chem Rev. 2018;118(3):1092–136. https://doi.org/10.1021/acs.chemrev.7b00122.
Lin H, Caroll KS. Introduction: posttranslational protein modification. Chem Rev. 2018;118(3):887–8. https://doi.org/10.1021/acs.chemrev.7b00756.
Perina D, Mikoč A, Ahel J, Ćetković H, Žaja R, Ahel I. Distribution of protein poly (ADP-ribosyl) ation systems across all domains of life. DNA Repair (Amst). 2014;23:4–16.
Chang P, Coughlin M, Mitchison TJ. Tankyrase-1 polymerization of poly (ADP-ribose) is required for spindle structure and function. Nat Cell Biol. 2005;7(11):1133–9. https://doi.org/10.1038/ncb1322.
Ozaki Y, Matsui H, Asou H, Nagamachi A, Aki D, Honda H, et al. Poly-ADP ribosylation of Miki by tankyrase-1 promotes centrosome maturation. Mol Cell. 2012;47(5):694–706. https://doi.org/10.1016/j.molcel.2012.06.033.
Hassa PO, Haenni SS, Elser M, Hottiger MO. Nuclear ADP-ribosylation reactions in mammalian cells: where are we today and where are we going? Microbiol Mol Biol Rev. 2006;70(3):789–829. https://doi.org/10.1128/MMBR.00040-05.
Gupte R, Liu Z, Kraus WL. PARPs and ADP-ribosylation: recent advances linking molecular functions to biological outcomes. Genes Dev. 2017;31(2):101–26. https://doi.org/10.1101/gad.291518.116.
Crawford K, Bonfiglio JJ, Mikoč A, Matic I, Ahel I. Specificity of reversible ADP-ribosylation and regulation of cellular processes. Crit Rev Biochem Mol Biol. 2018;53(1):64–82. https://doi.org/10.1080/10409238.2017.1394265.
Gibson BA, Kraus WL. New insights into the molecular and cellular functions of poly (ADP-ribose) and PARPs. Nat Rev Mol Cell Biol. 2012;13(7):411–24. https://doi.org/10.1038/nrm3376.
Vyas S, Matic I, Uchima L, Rood J, Zaja R, Hay RT, et al. Family-wide analysis of poly (ADP-ribose) polymerase activity. Nat Commun. 2014;5(1):4426. https://doi.org/10.1038/ncomms5426.
Hottiger MO, Hassa PO, Lüscher B, Schüler H, Koch-Nolte F. Toward a unified nomenclature for mammalian ADP-ribosyltransferases. Trends Biochem Sci. 2010;35(4):208–19. https://doi.org/10.1016/j.tibs.2009.12.003.
De Vos M, Schreiber V, Dantzer F. The diverse roles and clinical relevance of PARPs in DNA damage repair: current state of the art. Biochem Pharmacol. 2012;84(2):137–46. https://doi.org/10.1016/j.bcp.2012.03.018.
Pazzaglia S, Pioli C. Multifaceted Role of PARP-1 in DNA Repair and Inflammation: Pathological and Therapeutic Implications in Cancer and Non-Cancer Diseases. Cells. 2019;9(1). https://doi.org/10.3390/cells9010041.
Luo X, Kraus WL. On PAR with PARP: cellular stress signaling through poly (ADP-ribose) and PARP-1. Genes Dev. 2012;26(5):417–32. https://doi.org/10.1101/gad.183509.111.
Robert I, Karicheva O, Reina San Martin B, Schreiber V, Dantzer F. Functional aspects of PARylation in induced and programmed DNA repair processes: preserving genome integrity and modulating physiological events. Mol Asp Med. 2013;34(6):1138–52. https://doi.org/10.1016/j.mam.2013.02.001.
Rouleau M, Patel A, Hendzel MJ, Kaufmann SH, Poirier GG. PARP inhibition: PARP1 and beyond. Nat Rev Cancer. 2010;10(4):293–301. https://doi.org/10.1038/nrc2812.
Hoppe MM, Sundar R, Tan DSP, Jeyasekharan AD. Biomarkers for homologous recombination deficiency in Cancer. J Natl Cancer Inst. 2018;110(7):704–13. https://doi.org/10.1093/jnci/djy085.
Jackson SP, Bartek J. The DNA-damage response in human biology and disease. Nature. 2009;461(7267):1071–8. https://doi.org/10.1038/nature08467.
O'Connor MJ. Targeting the DNA damage response in Cancer. Mol Cell. 2015;60(4):547–60. https://doi.org/10.1016/j.molcel.2015.10.040.
Shibata A, Jeggo PA. DNA double-strand break repair in a cellular context. Clin Oncol (R Coll Radiol). 2014;26(5):243–9.
Ranjha L, Howard SM, Cejka P. Main steps in DNA double-strand break repair: an introduction to homologous recombination and related processes. Chromosoma. 2018;127(2):187–214. https://doi.org/10.1007/s00412-017-0658-1.
Scully R, Panday A, Elango R, Willis NA. DNA double-strand break repair-pathway choice in somatic mammalian cells. Nat Rev Mol Cell Biol. 2019;20(11):698–714. https://doi.org/10.1038/s41580-019-0152-0.
Symington LS. Mechanism and regulation of DNA end resection in eukaryotes. Crit Rev Biochem Mol Biol. 2016;51(3):195–212. https://doi.org/10.3109/10409238.2016.1172552.
Helleday T. Pathways for mitotic homologous recombination in mammalian cells. Mutat Res. 2003;532(1–2):103–15. https://doi.org/10.1016/j.mrfmmm.2003.08.013.
Hegde ML, Hazra TK, Mitra S. Early steps in the DNA base excision/single-strand interruption repair pathway in mammalian cells. Cell Res. 2008;18(1):27–47. https://doi.org/10.1038/cr.2008.8.
Ström CE, Johansson F, Uhlén M, Szigyarto CA, Erixon K, Helleday T. Poly (ADP-ribose) polymerase (PARP) is not involved in base excision repair but PARP inhibition traps a single-strand intermediate. Nucleic Acids Res. 2011;39(8):3166–75. https://doi.org/10.1093/nar/gkq1241.
Brem R. Hall J.XRCC1 is required for DNA single-strand break repair in human cells. Nucleic Acids Res. 2005;33(8):2512–20. https://doi.org/10.1093/nar/gki543.
Caldecott KW, Aoufouchi S, Johnson P, Shall S. XRCC1 polypeptide interacts with DNA polymerase beta and possibly poly (ADP-ribose) polymerase, and DNA ligase III is a novel molecular 'nick-sensor' in vitro. Nucleic Acids Res. 1996;24(22):4387–94. https://doi.org/10.1093/nar/24.22.4387.
Leppard JB, Dong Z, Mackey ZB, Tomkinson AE. Physical and functional interaction between DNA ligase IIIalpha and poly (ADP-ribose) polymerase 1 in DNA single-strand break repair. Mol Cell Biol. 2003;23(16):5919–27. https://doi.org/10.1128/MCB.23.16.5919-5927.2003.
Wang M, Wu W, Wu W, Rosidi B, Zhang L, Wang H, et al. PARP-1 and Ku compete for repair of DNA double strand breaks by distinct NHEJ pathways. Nucleic Acids Res. 2006;34(21):6170–82. https://doi.org/10.1093/nar/gkl840.
Langelier MF, Riccio AA, Pascal JM. PARP-2 and PARP-3 are selectively activated by 5′ phosphorylated DNA breaks through an allosteric regulatory mechanism shared with PARP-1. Nucleic Acids Res. 2014;42(12):7762–75.
Franzese E, Centonze S, Diana A, Carlino F, Guerrera LP, Di Napoli M, et al. PARP inhibitors in ovarian cancer. Cancer Treat Rev. 2019;73:1–9. https://doi.org/10.1016/j.ctrv.2018.12.002.
Grundy GJ, Rulten SL, Arribas-Bosacoma R, Davidson K, Kozik Z, Oliver AW, et al. The Ku-binding motif is a conserved module for recruitment and stimulation of non-homologous end-joining proteins. Nat Commun. 2016;7(1):11242. https://doi.org/10.1038/ncomms11242.
Gupta R, Somyajit K, Narita T, Maskey E, Stanlie A, Kremer M, et al. DNA Repair Network Analysis Reveals Shieldin as a Key Regulator of NHEJ and PARP Inhibitor Sensitivity. Cell. 2018;173(4):972–988.e923.
Helleday T. The underlying mechanism for the PARP and BRCA synthetic lethality: clearing up the misunderstandings. Mol Oncol. 2011;5(4):387–93. https://doi.org/10.1016/j.molonc.2011.07.001.
Lord CJ, Ashworth A. PARP inhibitors: synthetic lethality in the clinic. Science. 2017;355(6330):1152–8. https://doi.org/10.1126/science.aam7344.
Murai J, Huang SY, Das BB, Renaud A, Zhang Y, Doroshow JH, et al. Trapping of PARP1 and PARP2 by clinical PARP inhibitors. Cancer Res. 2012;72(21):5588–99. https://doi.org/10.1158/0008-5472.CAN-12-2753.
D'Andrea AD. Mechanisms of PARP inhibitor sensitivity and resistance. DNA Repair (Amst). 2018;71:172–6.
Slade D. PARP and PARG inhibitors in cancer treatment. Genes Dev. 2020;34(5–6):360–94. https://doi.org/10.1101/gad.334516.119.
Iyer S, Suresh S, Guo D, Daman K, Chen JCJ, Liu P, et al. Precise therapeutic gene correction by a simple nuclease-induced double-stranded break. Nature. 2019;568(7753):561–5. https://doi.org/10.1038/s41586-019-1076-8.
Ceccaldi R, Liu JC, Amunugama R, Hajdu I, Primack B, Petalcorin MI, et al. Homologous-recombination-deficient tumours are dependent on Polθ-mediated repair. Nature. 2015;518(7538):258–62. https://doi.org/10.1038/nature14184.
Ahrabi S, Sarkar S, Pfister SX, Pirovano G, Higgins GS, Porter AC, et al. A role for human homologous recombination factors in suppressing microhomology-mediated end joining. Nucleic Acids Res. 2016;44(12):5743–57. https://doi.org/10.1093/nar/gkw326.
Farmer H, McCabe N, Lord CJ, Tutt AN, Johnson DA, Richardson TB, et al. Targeting the DNA repair defect in BRCA mutant cells as a therapeutic strategy. Nature. 2005;434(7035):917–21. https://doi.org/10.1038/nature03445.
O'Neil NJ, Bailey ML, Hieter P. Synthetic lethality and cancer. Nat Rev Genet. 2017;18(10):613–23. https://doi.org/10.1038/nrg.2017.47.
Ashworth A, Lord CJ. Synthetic lethal therapies for cancer: what's next after PARP inhibitors? Nat Rev Clin Oncol. 2018;15(9):564–76. https://doi.org/10.1038/s41571-018-0055-6.
Lee JM, Ledermann JA, Kohn EC. PARP inhibitors for BRCA1/2 mutation-associated and BRCA-like malignancies. Ann Oncol. 2014;25(1):32–40. https://doi.org/10.1093/annonc/mdt384.
Moschetta M, George A, Kaye SB, Banerjee S. BRCA somatic mutations and epigenetic BRCA modifications in serous ovarian cancer. Ann Oncol. 2016;27(8):1449–55. https://doi.org/10.1093/annonc/mdw142.
Perkhofer L, Schmitt A, Romero Carrasco MC, Ihle M, Hampp S, Ruess DA, et al. ATM deficiency generating genomic instability sensitizes pancreatic ductal adenocarcinoma cells to therapy-induced DNA damage. Cancer Res. 2017;77(20):5576–90. https://doi.org/10.1158/0008-5472.CAN-17-0634.
Caumanns JJ, Wisman GBA, Berns K, van der Zee AGJ, de Jong S. ARID1A mutant ovarian clear cell carcinoma: a clear target for synthetic lethal strategies. Biochim Biophys Acta Rev Cancer. 2018;1870(2):176–84. https://doi.org/10.1016/j.bbcan.2018.07.005.
McCabe N, Turner NC, Lord CJ, Kluzek K, Bialkowska A, Swift S, et al. Deficiency in the repair of DNA damage by homologous recombination and sensitivity to poly (ADP-ribose) polymerase inhibition. Cancer Res. 2006;66(16):8109–15. https://doi.org/10.1158/0008-5472.CAN-06-0140.
Mirza MR, Monk BJ, Herrstedt J, Oza AM, Mahner S, Redondo A, et al. Niraparib maintenance therapy in platinum-sensitive, recurrent ovarian Cancer. N Engl J Med. 2016;375(22):2154–64. https://doi.org/10.1056/NEJMoa1611310.
Shen J, Zhao W, Ju Z, Wang L, Peng Y, Labrie M, et al. PARPi triggers the STING-dependent immune response and enhances the therapeutic efficacy of immune checkpoint blockade independent of BRCAness. Cancer Res. 2019;79(2):311–9. https://doi.org/10.1158/0008-5472.CAN-18-1003.
Lightfoot M, Montemorano L, Bixel K. PARP Inhibitors in Gynecologic Cancers: What Is the Next Big Development? Curr Oncol Rep. 2020;22(3):29.
Lin KY, Kraus WL. PARP Inhibitors for Cancer Therapy. Cell. 2017;169(2):183.
Bochum S, Berger S, Martens UM. Olaparib. Recent Results Cancer Res. 2018;211:217–33. https://doi.org/10.1007/978-3-319-91442-8_15.
Moore KN, Secord AA, Geller MA, Miller DS, Cloven N, Fleming GF, et al. Niraparib monotherapy for late-line treatment of ovarian cancer (QUADRA): a multicentre, open-label, single-arm, phase 2 trial. Lancet Oncol. 2019;20(5):636–48. https://doi.org/10.1016/S1470-2045(19)30029-4.
Coleman RL, Oza AM, Lorusso D, Aghajanian C, Oaknin A, Dean A, et al. Rucaparib maintenance treatment for recurrent ovarian carcinoma after response to platinum therapy (ARIEL3): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet. 2017;390(10106):1949–61. https://doi.org/10.1016/S0140-6736(17)32440-6.
Hoy SM. Talazoparib: First Global Approval. Drugs. 2018;78(18):1939–46. https://doi.org/10.1007/s40265-018-1026-z.
Koosha F, Neshasteh-Riz A, Takavar A, Eyvazzadeh N, Mazaheri Z, Eynali S, et al. The combination of A-966492 and Topotecan for effective radiosensitization on glioblastoma spheroids. Biochem Biophys Res Commun. 2017;491(4):1092–7. https://doi.org/10.1016/j.bbrc.2017.08.018.
Motta C, D'Angeli F, Scalia M, Satriano C, Barbagallo D, Naletova I, et al. PJ-34 inhibits PARP-1 expression and ERK phosphorylation in glioma-conditioned brain microvascular endothelial cells. Eur J Pharmacol. 2015;761:55–64. https://doi.org/10.1016/j.ejphar.2015.04.026.
Thorsell AG, Ekblad T, Karlberg T, Löw M, Pinto AF, Trésaugues L, et al. Structural basis for potency and promiscuity in poly (ADP-ribose) polymerase (PARP) and Tankyrase inhibitors. J Med Chem. 2017;60(4):1262–71. https://doi.org/10.1021/acs.jmedchem.6b00990.
Mizutani A, Yashiroda Y, Muramatsu Y, Yoshida H, Chikada T, Tsumura T, et al. RK-287107, a potent and specific tankyrase inhibitor, blocks colorectal cancer cell growth in a preclinical model. Cancer Sci. 2018;109(12):4003–14. https://doi.org/10.1111/cas.13805.
Bowman KJ, Newell DR, Calvert AH, Curtin NJ. Differential effects of the poly (ADP-ribose) polymerase (PARP) inhibitor NU1025 on topoisomerase I and II inhibitor cytotoxicity in L1210 cells in vitro. Br J Cancer. 2001;84(1):106–12. https://doi.org/10.1054/bjoc.2000.1555.
Meng X, Song W, Deng B, Xing Z, Zhang W. 3-aminobenzamide, one of poly (ADP-ribose)polymerase-1 inhibitors, rescuesapoptosisin rat models of spinal cord injury. Int J Clin Exp Pathol. 2015;8(10):12207–15.
Calabrese CR, Almassy R, Barton S, Batey MA, Calvert AH, Canan-Koch S, et al. Anticancer chemosensitization and radiosensitization by the novel poly (ADP-ribose) polymerase-1 inhibitor AG14361. J Natl Cancer Inst. 2004;96(1):56–67. https://doi.org/10.1093/jnci/djh005.
Cao R. Free energy calculation provides insight into the action mechanism of selective PARP-1 inhibitor. J Mol Model. 2016;22(4):74. https://doi.org/10.1007/s00894-016-2952-x.
Sharif-Askari B, Amrein L, Aloyz R, Panasci L. PARP3 inhibitors ME0328 and olaparib potentiate vinorelbine sensitization in breast cancer cell lines. Breast Cancer Res Treat. 2018;172(1):23–32. https://doi.org/10.1007/s10549-018-4888-6.
Lin X, Chen D, Zhang C, Zhang X, Li Z, Dong B, et al. Augmented antitumor activity by olaparib plus AZD1775 in gastric cancer through disrupting DNA damage repair pathways and DNA damage checkpoint. J Exp Clin Cancer Res. 2018;37(1):129. https://doi.org/10.1186/s13046-018-0790-7.
Chen K, Yang D, Li X, Sun B, Song F, Cao W, et al. Mutational landscape of gastric adenocarcinoma in Chinese: implications for prognosis and therapy. Proc Natl Acad Sci U S A. 2015;112(4):1107–12. https://doi.org/10.1073/pnas.1422640112.
Sahasrabudhe R, Lott P, Bohorquez M, Toal T, Estrada AP, Suarez JJ, et al. Germline Mutations in PALB2, BRCA1, and RAD51C, Which Regulate DNA Recombination Repair, in Patients With Gastric Cancer. Gastroenterology. 2017;152(5):983–986.e986.
Pyriochou A, Olah G, Deitch EA, Szabó C, Papapetropoulos A. Inhibition of angiogenesis by the poly (ADP-ribose) polymerase inhibitor PJ-34. Int J Mol Med. 2008;22(1):113–8.
Lacal PM, Tentori L, Muzi A, Ruffini F, Dorio AS, Xu W, et al. Pharmacological inhibition of poly (ADP-ribose) polymerase activity down-regulates the expression of syndecan-4 and Id-1 in endothelial cells. Int J Oncol. 2009;34(3):861–72. https://doi.org/10.3892/ijo_00000213.
Rajesh M, Mukhopadhyay P, Godlewski G, Bátkai S, Haskó G, Liaudet L, et al. Poly (ADP-ribose) polymerase inhibition decreases angiogenesis. Biochem Biophys Res Commun. 2006;350(4):1056–62. https://doi.org/10.1016/j.bbrc.2006.09.160.
Zampieri M, Guastafierro T, Calabrese R, Ciccarone F, Bacalini MG, Reale A, et al. ADP-ribose polymers localized on Ctcf-Parp1-Dnmt1 complex prevent methylation of Ctcf target sites. Biochem J. 2012;441(2):645–52. https://doi.org/10.1042/BJ20111417.
Kim CG, Lee H, Gupta N, Ramachandran S, Kaushik I, Srivastava S, et al. Role of Forkhead box class O proteins in cancer progression and metastasis. Semin Cancer Biol. 2018;50:142–51. https://doi.org/10.1016/j.semcancer.2017.07.007.
Park SH, Jang KY, Kim MJ, Yoon S, Jo Y, Kwon SM, et al. Tumor suppressive effect of PARP1 and FOXO3A in gastric cancers and its clinical implications. Oncotarget. 2015;6(42):44819–31. https://doi.org/10.18632/oncotarget.6264.
Yang XB, Zhao JJ, Huang CY, Wang QJ, Pan K, Wang DD, et al. Decreased expression of the FOXO3a gene is associated with poor prognosis in primary gastric adenocarcinoma patients. PLoS One. 2013;8(10):e78158.
Kubota E, Williamson CT, Ye R, Elegbede A, Peterson L, Lees-Miller SP, et al. Low ATM protein expression and depletion of p53 correlates with olaparib sensitivity in gastric cancer cell lines. Cell Cycle. 2014;13(13):2129–37. https://doi.org/10.4161/cc.29212.
Min A, Im SA, Yoon YK, Song SH, Nam HJ, Hur HS, et al. RAD51C-deficient cancer cells are highly sensitive to the PARP inhibitor olaparib. Mol Cancer Ther. 2013;12(6):865–77. https://doi.org/10.1158/1535-7163.MCT-12-0950.
Arora S, Balasubramaniam S, Zhang H, Berman T, Narayan P, Suzman D, et al. FDA Approval Summary: Olaparib Monotherapy or in Combination with Bevacizumab for the Maintenance Treatment of Patients with Advanced Ovarian Cancer. Oncologist. 2021;26(1):e164–72. https://doi.org/10.1002/onco.13551.
Yang L, Yang G, Ding Y, Huang Y, Liu S, Zhou L, et al. Combined treatment with PI3K inhibitor BKM120 and PARP inhibitor olaparib is effective in inhibiting the gastric cancer cells with ARID1A deficiency. Oncol Rep. 2018;40(1):479–87. https://doi.org/10.3892/or.2018.6445.
Amin O, Beauchamp MC, Nader PA, Laskov I, Iqbal S, Philip CA, et al. Suppression of homologous recombination by insulin-like growth factor-1 inhibition sensitizes cancer cells to PARP inhibitors. BMC Cancer. 2015;15(1):817. https://doi.org/10.1186/s12885-015-1803-y.
Rasmussen RD, Gajjar MK, Jensen KE, Hamerlik P. Enhanced efficacy of combined HDAC and PARP targeting in glioblastoma. Mol Oncol. 2016;10(5):751–63. https://doi.org/10.1016/j.molonc.2015.12.014.
Zhong L, Wang R, Wang Y, Peng S, Ma Y, Ding S, et al. Dual inhibition of VEGF and PARP suppresses KRAS-mutant colorectal cancer. Neoplasia. 2020;22(9):365–75. https://doi.org/10.1016/j.neo.2020.06.001.
Koustas E, Karamouzis MV, Sarantis P, Schizas D, Papavassiliou AG. Inhibition of c-MET increases the antitumour activity of PARP inhibitors in gastric cancer models. J Cell Mol Med. 2020;24(18):10420–31. https://doi.org/10.1111/jcmm.15655.
Barazzuol L, Jena R, Burnet NG, Meira LB, Jeynes JC, Kirkby KJ, et al. Evaluation of poly (ADP-ribose) polymerase inhibitor ABT-888 combined with radiotherapy and temozolomide in glioblastoma. Radiat Oncol. 2013;8(1):65. https://doi.org/10.1186/1748-717X-8-65.
Thomas HD, Calabrese CR, Batey MA, Canan S, Hostomsky Z, Kyle S, et al. Preclinical selection of a novel poly (ADP-ribose) polymerase inhibitor for clinical trial. Mol Cancer Ther. 2007;6(3):945–56. https://doi.org/10.1158/1535-7163.MCT-06-0552.
Bang YJ, Im SA, Lee KW, Cho JY, Song EK, Lee KH, et al. Randomized, double-blind phase II trial with prospective classification by ATM protein level to evaluate the efficacy and tolerability of Olaparib plus paclitaxel in patients with recurrent or metastatic gastric Cancer. J Clin Oncol. 2015;33(33):3858–65. https://doi.org/10.1200/JCO.2014.60.0320.
Bang YJ, Xu RH, Chin K, Lee KW, Park SH, Rha SY, et al. Olaparib in combination with paclitaxel in patients with advanced gastric cancer who have progressed following first-line therapy (GOLD): a double-blind, randomised, placebo-controlled, phase 3 trial. Lancet Oncol. 2017;18(12):1637–51. https://doi.org/10.1016/S1470-2045(17)30682-4.
Smyth E. Missing a GOLDen opportunity in gastric cancer. Lancet Oncol. 2017;18(12):1561–3. https://doi.org/10.1016/S1470-2045(17)30719-2.
Berlin J, Ramanathan RK, Strickler JH, Subramaniam DS, Marshall J, Kang YK, et al. A phase 1 dose-escalation study of veliparib with bimonthly FOLFIRI in patients with advanced solid tumours. Br J Cancer. 2018;118(7):938–46. https://doi.org/10.1038/s41416-018-0003-3.
Bindra RS, Gibson SL, Meng A, Westermark U, Jasin M, Pierce AJ, et al. Hypoxia-induced down-regulation of BRCA1 expression by E2Fs. Cancer Res. 2005;65(24):11597–604.
Friedlander M, Meniawy T, Markman B, Mileshkin L, Harnett P, Millward M, et al. Pamiparib in combination with tislelizumab in patients with advanced solid tumours: results from the dose-escalation stage of a multicentre, open-label, phase 1a/b trial. Lancet Oncol. 2019;20(9):1306–15. https://doi.org/10.1016/S1470-2045(19)30396-1.
Buchtel KM, Vogel Postula KJ, Weiss S, Williams C, Pineda M. Weissman SM.FDA approval of PARP inhibitors and the impact on genetic counseling and genetic testing practices. J Genet Couns. 2018;27(1):131–9. https://doi.org/10.1007/s10897-017-0130-7.
Lim JSJ, Tan DSP. Understanding Resistance Mechanisms and Expanding the Therapeutic Utility of PARP Inhibitors. Cancers (Basel). 2017;9(8). https://doi.org/10.3390/cancers9080109.
Acknowledgements
Not applicable.
Funding
This work was supported by grants from the Colorectal Cancer Medical Seeds Research Fund (2019095).
Author information
Authors and Affiliations
Contributions
YW and KZ conceived the structure of the manuscript; YW retrieved the relevant paper and finished the manuscript including figures and tables; YW, YH, HX, JS, RC and YZ revised the manuscript. All authors have contributed to the preparation of this manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Wang, Y., Zheng, K., Huang, Y. et al. PARP inhibitors in gastric cancer: beacon of hope. J Exp Clin Cancer Res 40, 211 (2021). https://doi.org/10.1186/s13046-021-02005-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s13046-021-02005-6